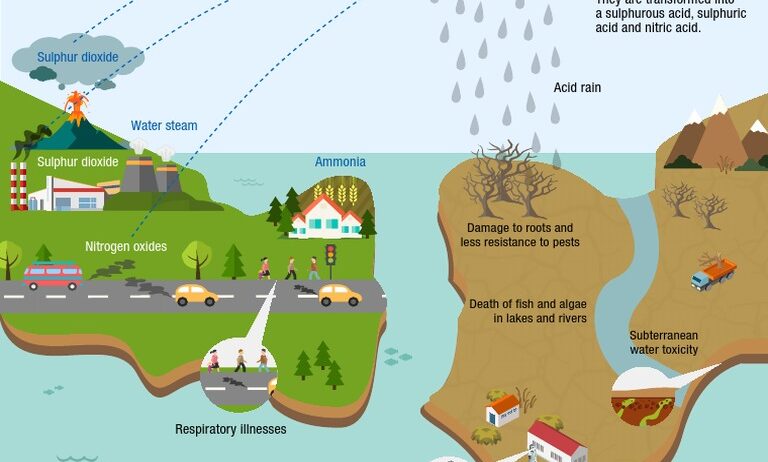
Acid rain is not just a casual aberration; it is a significant environmental menace that manifests as precipitation with a pH level lower than the natural range. The prevailing discourse on acid rain predominantly centers around its deleterious effects on the environment. However, to grasp the gravity of this phenomenon, it is imperative to delve into its primary contributors. This exposition seeks to elucidate the major causes of acid rain while emphasizing the intricate interconnections that underlie this environmental quandary.
At the crux of the acid rain issue lies a series of anthropogenic activities that introduce abnormal concentrations of sulfur dioxide (SO₂) and nitrogen oxides (NOₓ) into the atmosphere. These oxides react with water vapor, ultimately engendering sulfuric and nitric acids when they interact with atmospheric moisture. The genesis of these gases can be traced primarily to the combustion of fossil fuels, industrial emissions, and vehicular exhaust. Consequently, it is essential to dissect each contributor in detail to encapsulate the multifaceted nature of acid rain.
1. Fossil Fuel Combustion
The burning of fossil fuels for energy generation serves as one of the predominant sources of sulfur dioxide and nitrogen oxides. Power plants, especially those that operate on coal, natural gas, and oil, release copious amounts of sulfur dioxide into the atmosphere. As coal combustion releases sulfur from the coal itself, this process is particularly notorious for its contributions to acid rain. Moreover, the thermal power generation industry, dependent largely on fossil fuel combustion, is often situated in proximity to urban areas, exacerbating the problem through increased pollution levels in densely populated regions.
2. Industrial Emissions
Industrial facilities are not mere spectators in the acid rain drama; rather, they play a pivotal role as contributors. Factories that process ores, cement plants, and refineries emit sulfur dioxide and nitrogen oxides during their operations. Furthermore, the production of metals, such as copper and lead, generates fumes that compound the existing pollution levels in the atmosphere. Regulatory loopholes and lax enforcement of environmental laws in some regions can heighten this phenomenon, allowing industries to unleash harmful emissions without stringent oversight.
3. Automobiles and Transportation
Transportation is integral to modern civilization but is also a dire contributor to acid rain. Cars and trucks produce nitrogen oxides as a byproduct of fuel combustion while driving on the roads. Urban sprawl has led to an unprecedented increase in the number of vehicles on the road, compounding the problem. The emissions from transportation, when combined with industrial pollutants, create a toxic amalgamate, synergistically enhancing the potency of acid rain. In densely populated ecosystems, this phenomenon can wreak havoc on water bodies, soils, and vegetation in the vicinity.
4. Natural Sources
While anthropogenic factors are often in the spotlight, one must not overlook natural contributors to acid rain. Volcanic eruptions, forest fires, and lightning strikes can introduce sulfur dioxide and nitrogen oxides into the atmosphere. These natural events are not as frequent but can result in localized spikes in acid precipitation, particularly near volcanic regions. Understanding the natural contribution is essential, as it reminds us that the environment itself can generate pollutants, albeit at different scales and magnitudes compared to human activities.
5. Agricultural Practices
Agriculture, an indispensable domain for human sustenance, paradoxically harbors an ulterior motive when discussing acid rain. The use of nitrogen-based fertilizers leads to the emission of ammonia, which can react in the atmosphere to form nitric acid. Furthermore, livestock farming contributes to emissions of methane and ammonia, resulting in elevated nitrogen compounds in the air. This discrepancy underscores the need for sustainable agricultural practices that mitigate pollution while ensuring food security.
6. Deforestation and Land Use Changes
The human penchant for land conversion, deforestation, and urbanization can significantly exacerbate acid rain. Trees and vegetation act as natural counterbalances to air pollution. When trees are cut down or land is altered, the buffering capacity diminishes, and the pollutant concentrations rise. This alteration not only harms the immediate environment but also has far-reaching consequences on local ecosystems. A landscape bereft of trees is more susceptible to pollutants, resulting in enhanced acid rain effects on soil and waterways.
Through the meticulous examination of these contributors, it becomes evident that acid rain is an intricate tapestry interwoven with both human and natural influences. Addressing this pressing environmental issue necessitates a holistic understanding of its root causes. Solutions must encompass technological advancements, stringent regulations on emissions, and a paradigm shift towards sustainable energy sources.
The conversation surrounding acid rain extends beyond mere environmental ramifications; it implicates human health, biodiversity, and economic stability. As awareness grows, so too does the imperative to act. The collective onus falls on individual consumers, corporations, and governments alike to reconfigure our interactions with the planet. By understanding the multifaceted causes of acid rain, society can begin to mitigate its impacts and pave the way for a healthier and more sustainable future.